Ethics · Health · November 2025 · 10 min
Yazen Health, Gemma Collins and the ASA: when health marketing crosses the line
A 2025 influencer campaign for a prescription-only weight-loss service, banned by the ASA — and what it reveals about ethics, regulation and public health.

In an era of proactive health consumerism, digital marketing is a powerful tool for connecting people with information and services. This raises a critical question for practitioners: in what ways can marketing communications be ethical, especially in sensitive sectors like health? This essay will explore this question by analysing a 2025 social media campaign from digital health company Yazen Health and media personality Gemma Collins. The campaign promoted a prescription-only weight-loss service and was subsequently banned by the UK's Advertising Standards Authority (Advertising Standards Authority | Committee of Advertising Practice, 2025c), providing a clear case study of marketing that did not align with industry codes of practice.
gemmacollins ITS the GC here and I've got something to share with you today. I'm starting this year two sizes down, thanks to Yazen's weight loss app and medication.
It has absolutely changed my life. I've been working with Yazen — the digital healthcare provider — to manage my weight responsibly over the last two months. I feel healthier, I feel happier. I don't have that constant noise in my head 'You need to eat. You need to eat.'
Yazen was founded by doctors to support people struggling with obesity and weight-related issues. They use approved medication alongside support from their team of doctors, psychologists, dietitians, physiotherapists and your own YazenCoach, helping their patients take control of their health.
I'm so excited for anyone that has struggled like me for years. This has been life changing for me. I hope we can all embark on this journey together.
It's important to remember everyone's weight loss journey is personal and unique and you can talk to your GP if you're concerned!

The paid-for Instagram post featured a video where Collins discussed the service, stating: 'I'm not telling anyone to go on this medication, but it is prescribed on the NHS' (Advertising Standards Authority | Committee of Advertising Practice, 2025c). The ASA ruled the ad illegally promoted a prescription-only medicine (POM) to the public, a finding amplified by media outlets including the BBC and The Independent (Edwards, 2025; Clarke, 2025). This essay will analyse the campaign's ethical standing by considering its adherence to rules, real-world outcomes, stakeholder impact, and the professional judgement it demonstrated. It will also consider whether current regulations are fit for purpose in a new era of healthcare consumerism before offering practical recommendations for ethical marketing practice.
Understanding the Environment
PESTLE
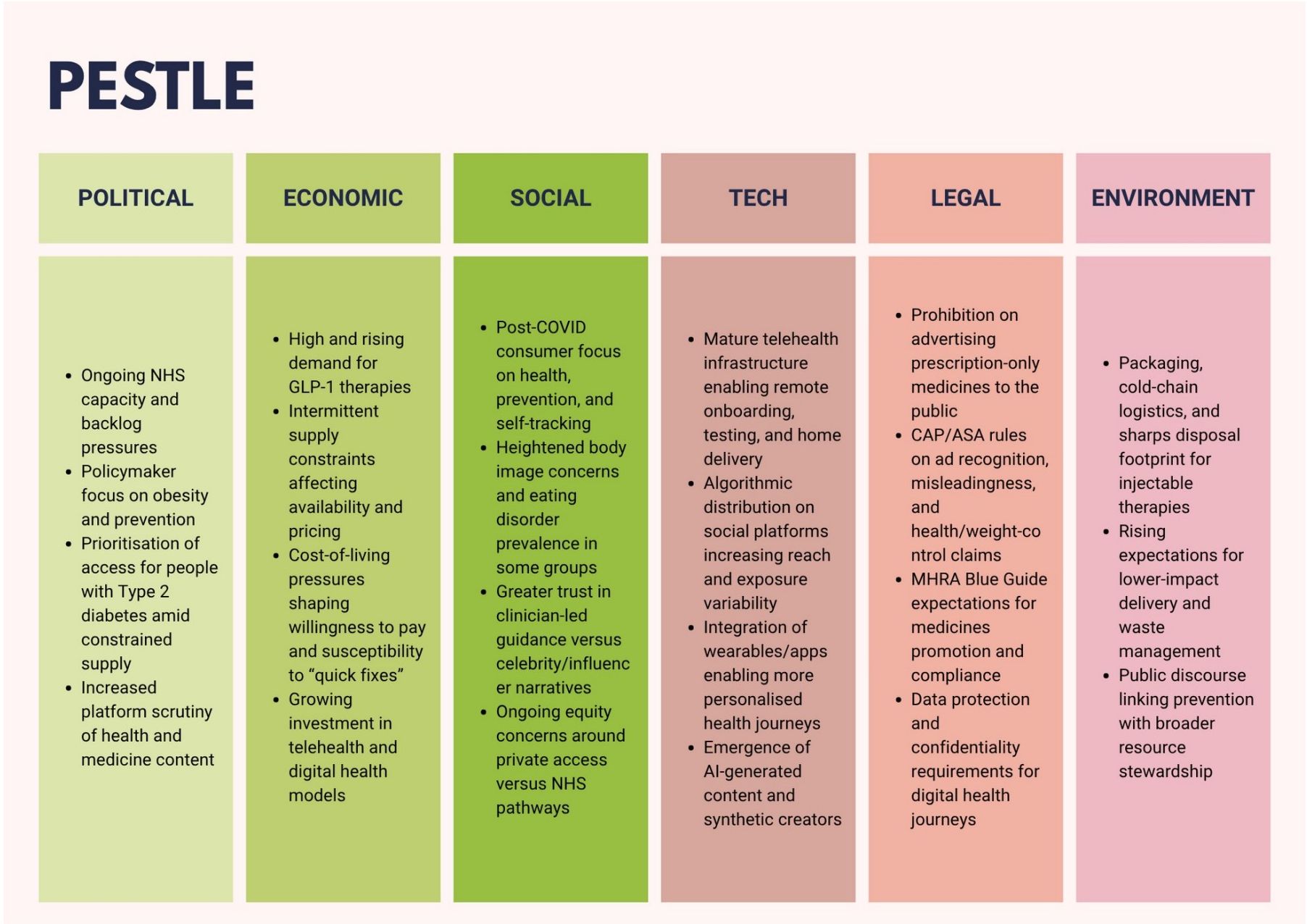
As seen in figure 3 above, since the pandemic, consumers have become more active in managing their health, driving demand for personalised care and digital services (Department of Health and Social Care, 2022). This trend coincides with a difficult economic backdrop, where a fracturing middle class faces mounting financial pressures, making them more anxious and value-conscious (WARC Exclusive, 2025a). At the same time, NHS capacity is under extreme pressure, making it difficult to get routine appointments and forcing primary care to prioritise patients with the highest clinical need (NHS England, 2024).
For weight management, the NHS recommends lifestyle changes first, reserving POMs like GLP-1 agonists for patients meeting strict clinical criteria (NHS, 2023). Private clinics like Yazen often have lower entry thresholds, widening access and driving demand. This has created significant market externalities. The rise of GLP-1s is already reshaping consumer buying habits, with analysts predicting volume declines for categories like confectionery and alcohol as users' calorie intake and cravings reduce (Barry, 2025; Whiteside, 2024). This phenomenon of 'want-loss' challenges the foundations of impulse-driven marketing, suggesting a future where substance and utility must replace sensory stimulation (Williams, 2025).
This marketing-fuelled demand also puts pressure on a strained healthcare system. GPs face an increased workload managing enquiries for medicines that are often in short supply, as flagged in national patient safety alerts (NHS England, 2024). This occurs within a strict regulatory framework where advertising POMs to the public is illegal (GOV.UK, 2025). This prohibition extends to influencer marketing, which must also meet standards on recognisability and social responsibility (Advertising Standards Authority | Committee of Advertising Practice, 2025a). However, the environment is complicated by the spread of health misinformation online by so-called 'quackfluencers,' which erodes public trust in formal healthcare systems (WARC, 2025c).
Campaign Analysis and Critique
The campaign's central failure was its direct breach of UK advertising law. The Instagram post promoted a POM to the public, an act forbidden by Rule 12.12 of the UK advertising code. The industry had been explicitly warned against this; a CAP enforcement notice, updated in April 2025, made clear that such ads were unlawful and must be removed (Advertising Standards Authority | Committee of Advertising Practice, 2025b). The ASA's subsequent ruling confirmed that phrases like 'weight loss medications' and the reference to the NHS would lead the public to understand the ad was for a POM. Collins's disclaimer did not mitigate this impression (Advertising Standards Authority | Committee of Advertising Practice, 2025c).
This failure demonstrated poor professional judgement and a disregard for the company's duty to follow clear legal and professional rules. Its real-world outcome was to stimulate public demand for a specific class of medicine, adding to the workload of GPs already under intense pressure (NHS England, 2024). The campaign had a negative impact on consumers, who were given a misleading picture of the treatment's availability, and on the NHS, which was left to manage the consequences.
Beyond its illegality, the promotion was socially irresponsible. Referencing the NHS misleadingly suggested a broad endorsement and easy access, creating a gap between public expectation and the clinical reality of strict NHS eligibility criteria (NHS, 2023). This undermines the principle of informed consent and risks patient safety by encouraging people to seek powerful medicines without a full clinical assessment. Given that influencer marketing on platforms like Instagram is effective because of its perceived authenticity and parasocial trust, a single high-profile post can rapidly influence a wide audience, including vulnerable groups (WARC Best Practice, 2025a).
Furthermore, the campaign had a damaging effect on the wider healthcare system. It drove patient enquiries for GLP-1 drugs during a period of national shortages, making it harder for patients with an established clinical need, like those with type 2 diabetes, to access their medication (Department of Health & Social Care, 2025). By prioritising a promotional message over its duty to be honest and responsible, the campaign put commercial goals ahead of public health and system stability.
From a purely commercial viewpoint, Yazen may have justified its campaign through several arguments. It could claim it was raising awareness of treatment options or positioning its service as a safer, regulated alternative to illicit online sellers. The marketing team may have simply misjudged the rules, believing an influencer's personal story with a disclaimer would be seen as educational, not promotional. Recent research shows consumers, especially 18–34s, rely heavily on creator content for health decisions, underscoring creators' high perceived credibility in sensitive categories (WARC Exclusive, 2025d).
However, these rationales are ethically weak. The legal duty to not advertise POMs is non-negotiable. The goal of attracting customers could have been achieved through compliant, condition-led campaigns that did not risk public safety or system stability. The potential harm to vulnerable consumers and the NHS was not adequately weighed against the commercial benefits of brand visibility. This reflects poor professional judgement, where clear risks were either downplayed or ignored in the pursuit of market share. ASA rulings consistently prioritise the effect of an ad over the advertiser's intent; if the content is understood to be promoting a POM, it breaches the rules (Advertising Standards Authority | Committee of Advertising Practice, 2025c).
Conclusion
The Yazen and Gemma Collins campaign serves as a clear example of how even well-intentioned efforts can have unintended, negative consequences when they do not align with ethical and regulatory standards. While Yazen and Collins may have sought to raise awareness of weight-loss options, promote Yazen's services, and contribute to the national conversation on tackling obesity, their campaign ultimately broke established guidelines and rules. By directly promoting prescription-only medicines and implying NHS endorsement, they encouraged members of the public to sign up without proper understanding of the potential side effects, whether they were clinically eligible, or what the short- and long-term implications of taking a prescription drug could be. Many people may have been reassured by seeing 'prescribed on the NHS,' without realising the strict criteria and medical supervision such treatments require.
Instead of supporting improved health literacy, the campaign risked misleading people and contributed to unnecessary pressure on already stretched NHS services. The targeted nature of the messaging, particularly during a time of heightened publicity around GLP-1 drugs, meant that vulnerable groups, including young adults, were at greater risk of harm, including negative effects on body image. This approach does not serve public health interests in the long term.
None of this is to say that regulations should never adapt. In a digital era where consumers are proactive about their health, there is value in more public awareness and understanding of prescription medicines. Thoughtful changes could help people make informed decisions and approach healthcare appointments with better knowledge of their conditions and possible treatments, crucial when NHS capacity is under strain. However, any future campaigns must remain firmly ethical and rooted in the protection of public health, ensuring transparency, evidence-based education, and safeguarding against harm, especially for vulnerable populations.
— Bex Sutton